Monday December 5, 2022
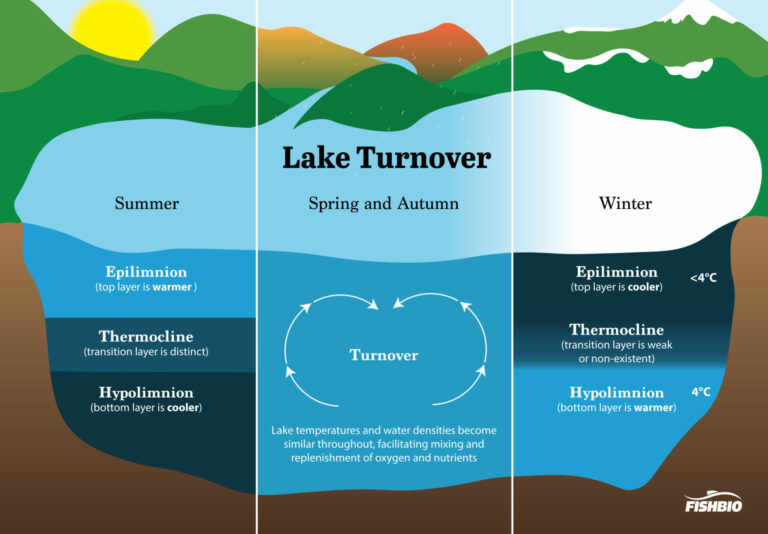
As fall sets in and the leaves begin to change, so does the water chemistry in local lakes and reservoirs. Water and air get colder, and sometimes smelly – and not only from decaying salmon carcasses. The foul, rotten-egg odor that many notice near lakes, reservoirs, and rivers during the fall is the result of a natural cycle of decay, a process dubbed “lake turnover.” In the summer, less dense warm water creates a surface layer referred to as the epilimnion. Cold water is dense and sinks to the bottom, forming a layer known as the hypolimnion. The thermocline, a thin transition layer of water, separates the epilimnion and hypolimnion and prevents the mixing of these two layers. This process of lake separation, known as stratification, typically only occurs in deep lakes greater than 20 ft deep and over the course of the summer months. In autumn, when cool weather lowers the temperature of the epilimnion, the thermocline begins to break down, and the epilimnion and the hypolimnion layers mix, a process that is often assisted by increasing winds and rain. In winter, the layers may separate again. In lakes where temperatures drop below freezing, the separation of layers is profound, with warmer water near the bottom and colder water on top. Once the surface water temperature drops to 4℃, the temperature at which fresh water is most dense, the top layer sinks to the bottom of the lake. As water above this layer continues to cool, the lake may begin to freeze, leaving a remaining layer of ice on the surface, because ice is less dense than water. However, in many California lakes, water temperatures will not drop this low. They will rarely ever freeze over, remaining stratified most of the year and only experiencing turnover during the winter months.

The phenomenon of lake turnover is essential for aquatic life, as it replenishes oxygen and nutrients throughout a body of water, but how is it connected to that rotten egg smell? Throughout the year, dead plants and other natural materials accumulate in the deep parts of a lake, where they decompose with assistance from bacteria that thrive in low-oxygen environments. As the bacteria break down these natural compounds, hydrogen sulfide gas (H2S) is released into the water. When the lake’s layers mix, the formerly deep water that contains more of this gas is brought to the surface, allowing the hydrogen sulfide to be released into the atmosphere. The same principle is responsible for the odor that sometimes occurs just downstream of dams, where water from the bottom of the reservoir is released. However, this odor usually disappears as the water is filled with oxygen by river turbulence as it flows downstream.

Lake turnover can occasionally lead to fish kills due to low oxygen levels in the hypolimnion layer. This may occur when a sudden thunderstorm or high wind rapidly mixes lake water, bringing oxygen-depleted water from the bottom to the surface, where most aquatic life is found. However, fish and other aquatic wildlife are typically able to avoid low oxygen zones by finding streams or springs that feed into the lake, or by moving to shallow areas where oxygen can dissolve easily. In extreme cases, however, lake turnover may happen too rapidly for fish to escape, especially in smaller lakes and ponds. In 2018, thousands of fish died in Lake Iroquois, a relatively small lake (less than one square mile in area) in central Illinois. The sudden deaths were likely due to a lake turnover event caused by an unexpected drop in temperature.
The odors that lake turnover often generates are not usually a cause for human health concerns. Hydrogen sulfide gas is quickly broken down in the environment and easily dispersed by wind. However, prolonged exposure can lead to nausea, headache, and fatigue. Although the human body can quickly process hydrogen sulfide, it is not recommended to drink out of bodies of water that have a rotten egg odor due to the possible presence of harmful bacteria. So if you encounter a stinky lake this fall, just know that it’s all part of nature’s nutrient recycling system.
This post was featured in our weekly e-newsletter, the Fish Report. You can subscribe to the Fish Report here.
